Professor Ferenc Krausz is an award-winning pioneer of attosecond physics, in which extremely short pulses of light are used primarily to gain a better understanding of the way electrons behave in atoms, molecules and solids. But he now wants to focus on something completely different: the early detection of common diseases worldwide. An interview.
The Interpreter: Ferenc Krausz on attosecond physics
Read more
Two years ago, you received the Nobel Prize for your pioneering work in a very specific area of physics, namely attosecond physics. Now you want to detect cancer and other diseases at an early stage. How do the two things fit together?
Ferenc Krausz: In a word: wonderfully. This shows firstly how important basic research is, but also how important it is in due course to identify applications that may emerge from this fundamental work – even if, as is the case with early detection, they may appear to be heading in a completely different direction.
Does this mean that attosecond physics has evolved?
It has at least gone through several phases. The fundamental work that ultimately led to the Nobel Prize being awarded was clearly driven very much by curiosity: We wanted to observe how electrons move at the atomic level and to do this we needed an extremely fast camera – our attosecond pulses. The second step we needed to make was to ensure that these new images actually showed what the theory predicted they would, so we had to validate the method. It was only after we’d done this that we could use our attosecond tools and measurement technologies to examine complex physical systems and discover new things.
And have all the questions now been answered?
No, far from it. Particularly in more complex systems, molecules and solids, there are still plenty of exciting questions that we might be able to help answer using our methods.
Despite this, you’re now keen to focus on an entirely different set of problems.
At some point, it was midway through the last decade, I asked myself the question: Do I really want to carry on doing what I’m currently researching right through to the end of my active career? Or am I still young enough to try something new? And the answer was very clear: I’ve still got enough energy and desire and motivation inside me to embark on the next step. So how about coming up with an application in which the tools that we’re experts at deploying in our group can be used to benefit the general public.
Attosecond physics in medicine? “I’ve still got enough energy and desire and motivation inside me to embark on the next step,” says Nobel laureate Ferenc Krausz.
© Daniel Gerst
And this is where healthcare came up as a possibility?
Yes, there were lots of discussions in our group, and in the end the decision we made was to use the attosecond measurement technology to tackle biomedical questions.
What are your plans exactly?
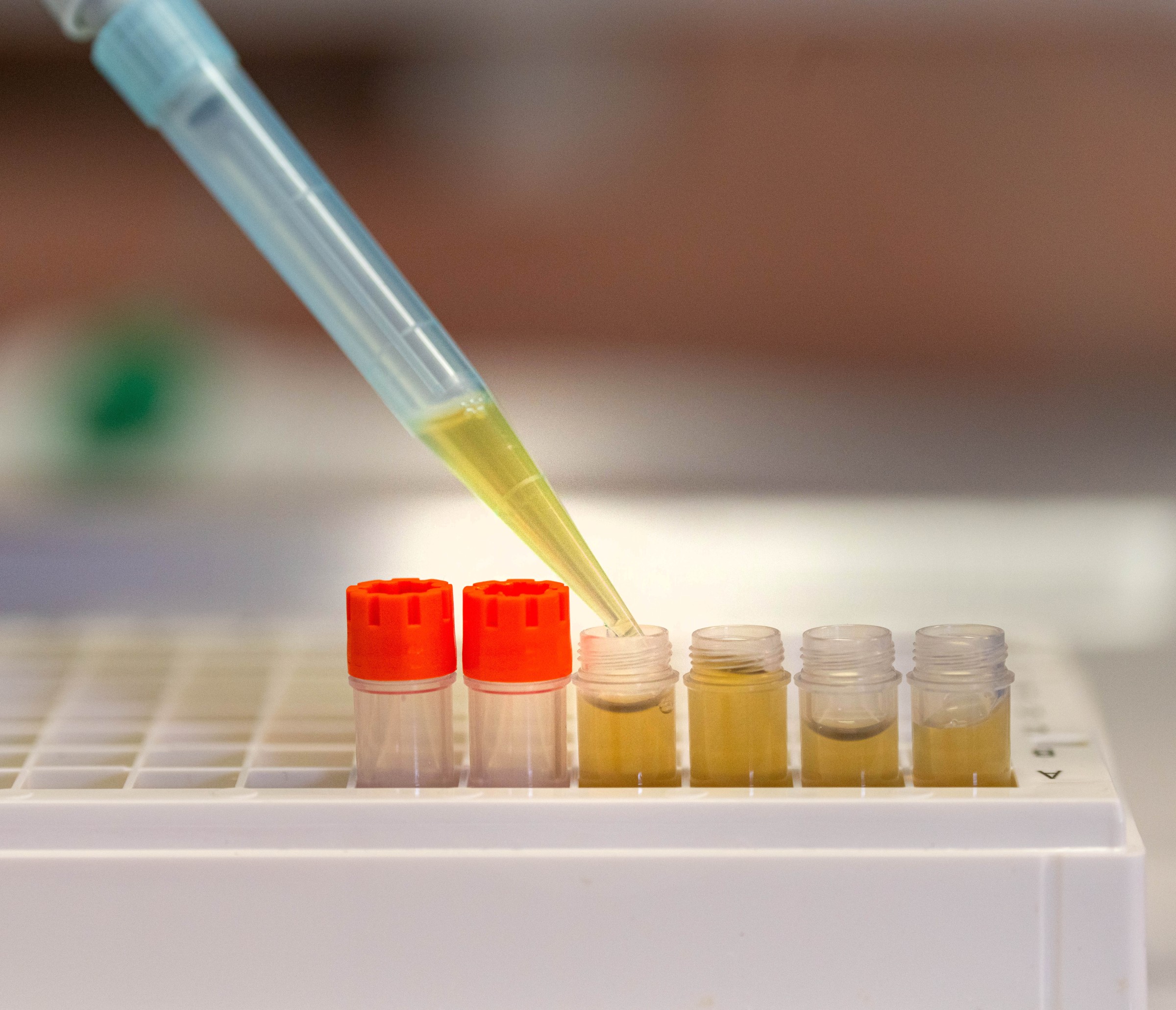
Our idea is that, if blood serum or blood plasma is suddenly irradiated with a very short, infrared laser pulse, then this will excite the molecules contained in the blood and cause them to pulse. These molecules will in turn emit infrared light, which we can accurately scan using our attosecond technology. This produces a kind of fingerprint. If a disease has altered the composition of the molecules in the blood, this should be apparent in the infrared fingerprint. This would make it possible to detect diseases at a very early stage.
We quickly realized that this new challenge would take us away from physics – toward chemistry, biochemistry and ultimately medicine. That’s why it was the right time to bring in appropriate expertise to enhance our group.
Ferenc Krausz
So, a group of physicists is suddenly keen to make an impact and succeed in medicine? This sounds, if I may say so, a little bold.
Well, we quickly realized that this new challenge would take us away from physics – toward chemistry, biochemistry and ultimately medicine. That’s why it was the right time to bring in appropriate expertise to enhance our group. And in Mihaela Žigman, we found a very talented colleague who would have had everything she needed to have a tremendous career in molecular biology even without engaging and working with physicists. But she was bold enough to embrace the challenge, as the only life scientist at the time, of joining a group that consisted more or less entirely of physicists and engineers. We were absolutely delighted. But the network within LMU was a big help as well.
In what way?
Our Munich Center for Advanced Photonics Cluster of Excellence at the time boasted a long and successful history of working with very well-known colleagues from the field of medicine – especially Professor Maximilian Reiser, the now emeritus director of radiology at Klinikum Großhadern. He was the first person we approached with our idea as we wanted to know his opinion. Max thought it was a good idea and he put us in touch with people directly: with Professor Christian Stief from the Urological Clinic, with Professor Nadia Harbeck, Director of the Breast Center, and with Professor Jürgen Behr, Director of Pneumology. All three of them were very open and extremely interested.
The idea: irradiate blood serum or blood plasma with a very short laser pulse, thereby causing the molecules in the blood to vibrate. The molecules then emit light that can be detected using attosecond technology. This creates a kind of fingerprint. "We’ve now collected so many samples of blood from test subjects and control groups that we’re now even able to identify individual stages of the cancers in our signals", says Ferenc Krausz.
© Thorsten Naeser
By working with these people, you covered three important diseases in the form of prostate cancer, breast cancer and lung cancer.
Correct, and for all the different types of cancer we’ve studied, we can see characteristic changes in the fingerprint of the blood that we can assign to these diseases. We’ve now collected so many samples of blood from test subjects and control groups that we’re now even able to identify individual stages of the cancers in our signals. This has given us a great deal of encouragement and motivation to carry on.
How exactly do you plan to carry on?
Rather than studying individual diseases, our focus is now on general healthcare. Our big aim is to create an individual infrared fingerprint for each person by taking repeated measurements, and to monitor how this fingerprint changes over time. Artificial intelligence should then help us to analyze these changes and identify diseases such as diabetes, heart or respiratory diseases – and we should be able to do this long before any symptoms occur or people go to see their doctor.
Rather than studying individual diseases, our focus is now on general healthcare. Our big aim is to create an individual infrared fingerprint for each person by taking repeated measurements, and to monitor how this fingerprint changes over time.
Ferenc Krausz
And is this realistic?
We’re currently doing this very thing in Hungary by conducting a study in which we intend to analyze the blood of 15,000 people twice a year over a period of at least ten years. But that’s not enough.
Do you need more people to volunteer as test subjects?
To obtain a truly meaningful set of data that will help create an early warning system, we need 50,000 to 100,000 participants, ideally from all over the world, to cover all the different ethnic groups. This is why in 2024 we came up with the plan to establish a global research alliance, which we’ve called Protecting.Health. I had the opportunity at the time to present the idea to Markus Blume, the Bavarian State Minister for Science and the Arts. He encouraged us to write a concept paper that ultimately even inspired Germany’s new federal government to champion preventive medicine. So the political will is there. We now need action to back this up.
“We need to view health as a global commons”
Read more
You’ve taken the step of moving on to Hong Kong.
As half of all humans on the planet live in Asia, we urgently need blood samples from this region. With the support of LMU, I’ve been able to take up a part-time professorship at the University of Hong Kong and lead a joint study there as part of Protecting.Health. The relevant contracts were signed in February, so we hope to be able to get started by the summer – which is much earlier than we’ve been able to in Germany. This should serve as a little motivation for the powers-that-be here at home to make a decision as soon as possible.
Is competition good for business?
The calculation is a very simple one: the agreement states that any possible income from the future marketing of a screening algorithm for the early detection of common diseases will be based on the contributions that are made to this. This means the sooner Germany starts collecting blood samples to train such an algorithm, the greater our share in its intellectual property will ultimately be – and future revenues will be bigger too.